The first question is, who do you test? If you’re using next-generation sequencing for nonsquamous carcinoma already, then on most of the panels, by default, you’re getting tumor mutational burden. What’s controversial is the optimal way to integrate it into clinical practice. I think we do have multiple data sets showing that it’s a legitimate predictive biomarker.
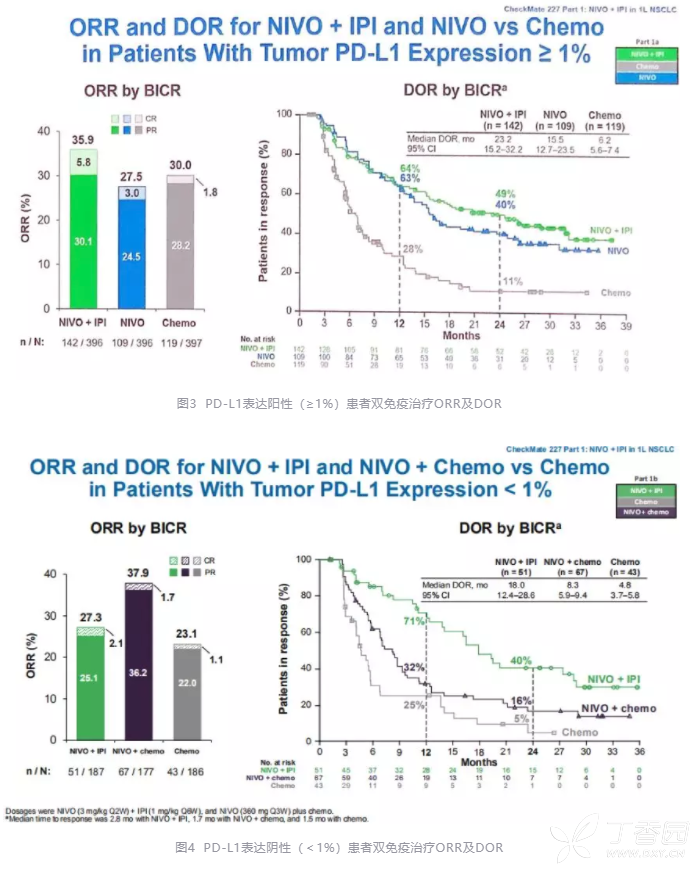
Jared Weiss, MD: TMB is a somewhat controversial biomarker. For those patients with high tumor mutation burden, regardless of their PD-L1 status, it does appear that they’re going to benefit from immunotherapy whether it’s the combination of ipilimumab/nivolumab, single agent PD-1, or a combination of chemotherapy and PD-1 inhibitor. There are a lot of patients who have no PD-L1 expression but have a high tumor mutation burden. But it turns out that it doesn’t overlap very cleanly. You might predict that those 2 biomarkers would overlap. One might presume that if you’ve heard PD-L1 predicts for patients who are going to respond, then you’ve also heard that patients with high TMB are going to respond. How these 2 biomarkers interact is, of course, critically important to understand. We’ve talked a lot about PD-L1 as a biomarker in patients with immunotherapy, but TMB, or tumor mutational burden, is also an important biomarker. That was significantly better than those patients who received chemotherapy alone. But in the group of patients who had high TMB, there was a clear correlation with benefit with ipilimumab and nivolumab. In CheckMate-227, there are certainly multiple arms of patients. I think regarding the subsets of patients who are PD-L1-negative or who have PD-L1 expression 1% or greater, among both groups it does appear that those patients seemed to benefit from ipilimumab and nivolumab if they had high TMB.

But it seems clear that patients with high tumor mutation burden seemed to benefit most from the combination of ipilimumab and nivolumab. I think over the course of 2 big scientific meetings recently, both AACR and ASCO, we’ve seen 2 different pieces of data presented. Ipilimumab is a CTLA-4 antibody and nivolumab is a PD-1 antibody.
Checkmate 227 plus#
There’s chemotherapy, chemotherapy plus nivolumab, and an arm of patients who got ipilimumab plus nivolumab.

Checkmate 227 trial#
The cutoff point in tumor mutation burden is 10 mutations per megabase.Īs the trial has gone forward, there are 3 arms in the trial in terms of treatment. The trial’s gone on to divide those patients further by their tumor mutation burden. The initial cutoff point for CheckMate-227 was patients who had PD-L1 expression greater than 1% or PD-L1 expression less than 1%. Riely, MD, PhD: CheckMate-227 was a clinical trial that enrolled patients with non-small cell lung cancer and has a variety of pieces of data that we’ve seen in several presentations.
